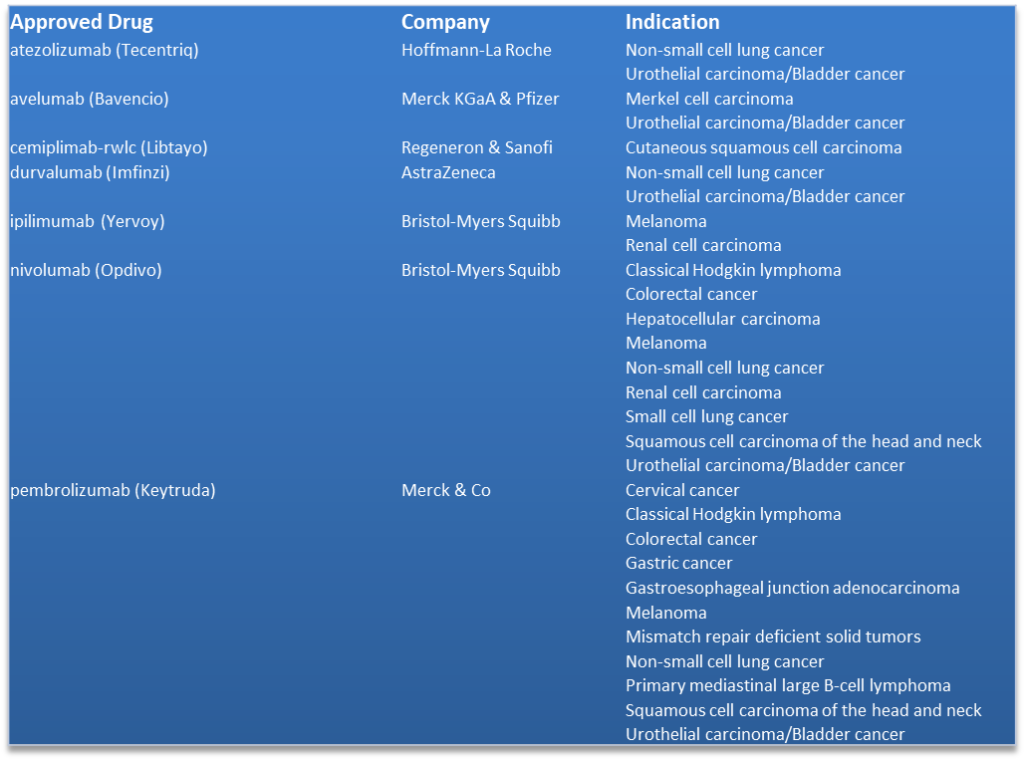
On October 1, 2018 Aduro Biotech, Inc. (NASDAQ: ADRO) reported that four separate abstracts from its research and development portfolio will be presented at the upcoming Society for Immunotherapy of Cancer (SITC) (Free SITC Whitepaper) annual meeting in Washington, D.C. from November 7-11, 2018 (Press release, Aduro Biotech, OCT 1, 2018, View Source;p=RssLanding&cat=news&id=2369686 [SID1234530081]). Preliminary clinical data from the ongoing Phase 1 dose-finding study evaluating ADU-S100 (MIW815), an intratumoral STING agonist in patients with advanced solid tumors or lymphomas, were accepted for presentation on November 9, 2018.
Schedule your 30 min Free 1stOncology Demo!
Discover why more than 1,500 members use 1stOncology™ to excel in:
Early/Late Stage Pipeline Development - Target Scouting - Clinical Biomarkers - Indication Selection & Expansion - BD&L Contacts - Conference Reports - Combinatorial Drug Settings - Companion Diagnostics - Drug Repositioning - First-in-class Analysis - Competitive Analysis - Deals & Licensing
Schedule Your 30 min Free Demo!
"We look forward to sharing preliminary clinical data from the dose escalation portion of the monotherapy trial which provides an initial understanding of the potential role of ADU-S100 in the treatment of cancer and which contribute to the broader scientific understanding of the STING pathway," said Stephen T. Isaacs, chairman, president and chief executive officer of Aduro Biotech. "In collaboration with our partner Novartis, our objective is to characterize the safety and mechanism of action of ADU-S100 across a wide array of solid tumors and lymphomas and provide a basis for continued development of ADU-S100 in combination with checkpoint inhibitors targeting PD-1 and CTLA-4."
Researchers will present additional preclinical data for ADU-S100 in combination with immune checkpoint inhibitors. They will also present the results of early research to identify and characterize an anti-SIRPa antibody ADU-1805.
Details of the poster and oral presentations are as follows:
Abstract 10763: Phase I dose-finding study of MIW815 (ADU-S100), an intratumoral STING agonist, in patients with advanced solid tumors or lymphomas
Date: November 9-10, 2018
Location: Poster Hall E, Walter E. Washington Convention Center
Abstract 10938: ADU-S100 (MIW815) Synergizes with Checkpoint Inhibition to Elicit an Anti-Tumor CD8+ T Cell Response to Control Distal Tumors
Date: November 9-10, 2018
Location: Poster Hall E, Walter E. Washington Convention Center
Abstract 10923: SIRPα blockade increases the activity of multiple myeloid lineage cells, enhances dendritic cell cross-presentation, and aids in remodeling the tumor microenvironment
Session: Rapid Oral Abstracts
Date/Time: November 9, 2018, 1:00 – 2:00 p.m. ET
Location: Poster Hall E, Walter E. Washington Convention Center
Abstract 10960: Pan-allele anti-SIRPα antibodies that block the SIRPα–CD47 innate immune checkpoint
Date: November 9-10, 2018
Location: Poster Hall E, Walter E. Washington Convention Center
About STING Pathway Activator Platform
The Aduro-proprietary STING pathway activator product candidates, including ADU-S100 (MIW815), are synthetic small molecule immune modulators that are designed to target and activate human STING. STING is generally expressed at high levels in immune cells, including dendritic cells. Natural activation of STING is not always sufficient to prevent the growth and spread of cancer cells. In preclinical models, ADU-S100 directly activates STING to further amplify the natural anti-tumor response. Once activated, the STING receptor initiates a profound innate immune response through multiple pathways, inducing the expression of a broad profile of cytokines, including interferons and chemokines. This subsequently leads to the development of a systemic tumor antigen-specific T cell adaptive immune response.
Aduro’s lead molecule, ADU-S100, is the first therapeutic in development specifically targeting STING. In collaboration with Novartis, it is being tested in a Phase 1 clinical trial as a single agent and in combination with ipilimumab, and in a Phase 1b combination trial with spartalizumab (PDR001), an investigational anti-PD-1 compound. These studies are enrolling patients with cutaneously accessible, advanced/metastatic solid tumors or lymphomas. The trials are evaluating the ability of ADU-S100 to activate the immune system and recruit specialized immune cells to attack the injected tumor, leading to a broad immune response that seeks out and kills distant metastases.